| LAUROYL PEROXIDE | ||
|
PRODUCT IDENTIFICATION |
||
| CAS NO. | 105-74-8 |
|
| EINECS NO. | 203-326-3 | |
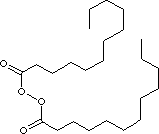
| FORMULA | (C11H23CO)2O2 | |
| MOL WT. | 398.63 | |
|
H.S. CODE |
2909.60 |
|
|
TOXICITY |
|
|
| SYNONYMS | Bis(1-oxododecyl) Peroxide; | |
| Dilauroyl Peroxide; Dodecanoyl peroxide; Peróxido de dilauroílo; Peroxyde de dilauroyle; Didodecanoyl peroxide; | ||
| SMILES | ||
|
CLASSIFICATION |
|
|
|
PHYSICAL AND CHEMICAL PROPERTIES |
||
| PHYSICAL STATE | White powder | |
|
MELTING POINT |
49 - 57 C | |
| BOILING POINT | ||
| SPECIFIC GRAVITY | ||
|
SOLUBILITY IN WATER |
Insoluble | |
| pH | ||
| VAPOR DENSITY | 13.7 | |
|
AUTOIGNITION |
112 C | |
| NFPA RATINGS | Special Hazard: OX | |
|
REFRACTIVE INDEX |
| |
| FLASH POINT | > 110 C | |
| STABILITY | This compound decomposes on exposure to heat and sunlight. | |
|
APPLICATIONS |
||
|
Peroxide: Compound containing the peroxy group (-O-O-), chainlike structure,
containing two oxygen atoms, each of which is bonded to the other and to a
radical or some element. It is considered that hydrogen peroxide is the starting
material to prepare organic and inorganic peroxides commercially. Hydrogen
Peroxide H2O2, is a powerful oxidizing agent. The most valuable property of
hydrogen peroxide is that it breaks down into water and oxygen and therefore
does not form any persistent, toxic residual compounds. It is used in the
processes of epoxidation, oxidation, hydroxylation and reduction. Its oxidizing
properties are used in the bleachings and deodorizing for textile, hair and in
paper manufacture. It is also used medicinally as an antiseptic. Its
application involves the production of chemicals like perhydrates as well as
organic peroxides in which some organic (or inorganic) substituents have
replaced one or both hydrogens. Some metals form peroxides in air sodium, barium
or zinc. Metal peroxide releases oxygen slowly in contact with atmospheric
moisture and used to as disinfectants in cosmetics, detergents, toothpaste and
pharmaceuticals. They can be used in the bleachings and deodorizing and a oxygen
release source in agricultural application to generate contaminated soils and
lakes. Organic Peroxides are powerful oxidizing agents releasing oxygen. They
are widely used as initiators,catalysts and crosslinking agent for the
polymerization process in the plastics manufacturing industry and as chemical
intermediates, bleaching agents, drying and cleaning agents. They are also used
as antiseptics, disinfectants and germicides medically for cosmetics,
detergents, toothpaste and pharmaceuticals. Organic peroxides are classified
in peroxydicarbonates, peroxyketals, peroxyesters, ketone
peroxides, hydroperoxides, dialkyl peroxides, diacyl peroxides by HMIS.
Lauroyl peroxide is a strong free radical source containing more than 4.0% of active oxygen; used as a polymerization initiator, catalyst and vulcanizing agent. The half-life temperatures are 62 C (for 10 hours), 80 C (1 for 1 hour) and 120 C (for 1 minute). It decomposes rapidly, causing fire and explosion hazard, on heating and under influence of light. It reacts violently with incompatible substances or ignition sources (acids, bases, reducing agents, and heavy metals). It should be stored in a dry and refrigerated (< 27C recommended or 38 C max) area and to keep away from reducing agents and incompatible substances. |
||
| SALES SPECIFICATION | ||
|
APPEARANCE |
white crystals | |
| PURITY |
99.0% min | |
| VOLATILE MATTER |
0.1% max | |
|
MELTING POINT |
49 - 57 C | |
| TRANSPORTATION | ||
| PACKING | 20kg in paper box | |
| HAZARD CLASS | 5.2 (Packing Group: II) | |
| UN NO. | 3106 (solution: 3109) | |
| OTHER INFORMATION | ||
| Hazard Symbols: O, Risk Phrases: 7-36/38-51/53, Safety Phrases: 3/7-14-36/37/39 | ||